The Hip Pocket

The Snapshot
- Pregnancy rates reduced by 23%. Calf loss increased by 9% – A recent study of factors affecting the reproductive performance of beef breeding herds found that where there was evidence of widespread and/or recent pestivirus infection, the percentage of lactating cows that became pregnant within four months of calving reduced by 23%, and calf wastage increased by 9%.
- 70% greater likelihood of developing Bovine Respiratory Disease (BRD)- Due to its suppressive effects on the immune system, cattle that are infected with pestivirus in feedlots have a 70% greater likelihood of developing Bovine Respiratory Disease (BRD) resulting in increased use of antibiotics in feedlots. Despite this, a cost: benefit of rectifying this situation and implementing mitigation strategies has not been determined.
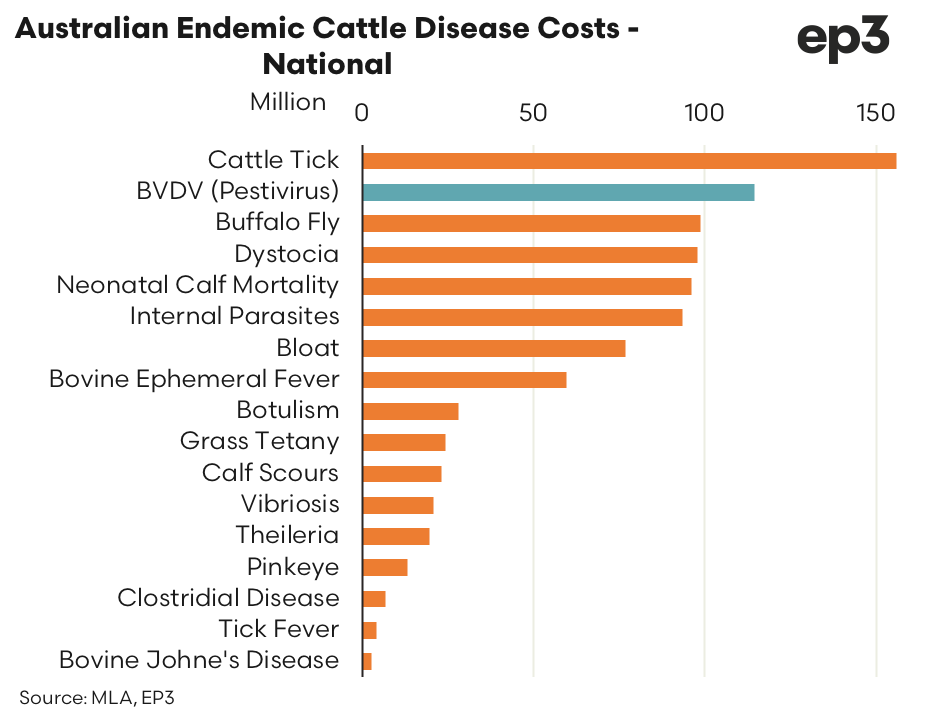
- $114 million per year in 2015 – Meat and Livestock Australia calculated the estimated cost of pestivirus in Australia was $114 million per year in 2015, only second to cattle tick. Australian cattle prices have doubled since that time.
The Detail
Episode3 (EP3), with the support of Zoetis Australia, have been investigating the impact of Bovine Viral Diarrhoea Virus (BVDV), also known as pestivirus, on the Australian cattle sector and this piece is the third instalment in a series of articles on pestivirus that will focus upon:
- What the virus is, how widely it is distributed and how it is spread (Published 14th October)
- The impact of the virus on cattle, types of infected animals, implications for breeding and current control measures (Published 28th October)
- The economic cost of the virus, impacts to the beef, feedlot and dairy sector
- The international response to the virus and the consideration of a control/eradication program for Australia
The cost of Pestivirus
There have been various models of the economic impact of pestivirus in Australia. All of these have been done with limited scope, such as a focus on the reproductive impacts within beef herds or dairy herds. Therefore, they underestimate the full impact of pestivirus on the national cattle herd and the individual farm arising from its impact on reproduction, calf wastage, increased animal health costs and increased antibiotic usage.
Additionally, the disruption of breeding conception/calving patterns has ramifications for productivity in that breeding season and subsequent seasons have not been accounted for in modelling of the cost of the virus.
For example:
- Empty cows or late calves have an opportunity cost and late conceptions will result in less time for recovery post calving in relation to the next joining.
- This can result in genetically sound or superior animals being culled because they were impacted by pestivirus rather than because they were genetically inferior.
- In turn, late calves have less time to grow out and will do so out of the optimal feed quality window resulting in lower end weights and higher cost of gain. In many systems, this results in farmers carrying weaners into the following season.
- This series of events results in increased cattle handling and labour inefficiencies compared with a timely, tighter calving.
- The knock-on effect is that heifers that have failed to achieve target mating weights are likely to join late, calve late and have lower lifetime beef production per breeding unit. No models to date have captured this effect in pestivirus economic impact.
In 2015 Meat and Livestock Australia (MLA) commissioned a report to assess the economic cost of a range of endemic diseases impacting the red meat sector. Pestivirus was included in this list as one of the costliest diseases facing the Australian cattle sector. However, this study didn’t include the impact of pestivirus on increased respiratory disease in feedlots.
The authors surveyed producers to create a list of diseases of concern and the economic cost was determined by assessing published evidence and where this was lacking using their professional judgement to estimate production losses, preventive costs and treatment costs associated with each disease.
The modelling included key variables associated with the economic cost of pestivirus such as virus prevalence, cattle mortality, reproductive loss, weight loss and costs associated with prevention or treatment of the disease. The survey results provided cost estimates at the herd level which were then extrapolated to a national basis, using herd demographics from the 2010-11 Australian Bureau of Statistics Agricultural Census.
The total cost of pestivirus in Australia was calculated at $114 million per year in 2015. The prevention costs of pestivirus were estimated at $7.7 million per year, nationally. The bulk of the cost of pestivirus was allocated to production losses and was calculated at $106.7 million per annum. On a national basis pestivirus was the second costliest disease facing the Australian cattle sector behind cattle tick at an estimated cost of $156 million per year
There were some limitations to the 2015 analysis given the fact that the modelling was based on the average cost per year rather than accounting for the cyclical nature of infectious disease. Models like this don’t highlight the consequence that infection of a susceptible herd can result in a business threatening loss for those producers who find themselves in this position.
A recent comprehensive study of factors affecting the reproductive performance of rangeland beef breeding herds in northern Australia found that where there was evidence of widespread and/or recent pestivirus infection (i.e. these were susceptible breeding females that were infected during the critical stage of pregnancy), the percentage of lactating cows that became pregnant within four months of calving was reduced by 23%, and calf wastage was increased by 9%.[1] It is well documented that events like this are likely to occur during periods of restocking or herd expansion when farmers are often purchasing breeding females at a premium.
Since the 2015 study the value of cattle in Australia has more than doubled. It would be logical to assume that, with the high prices for cattle, a virus that impacts the reproductive capacity of cattle and increases their susceptibility to other diseases would have a significantly higher economic cost.
Pestivirus on the dairy farm
There is an increased incidence of reporting and analysis of the impact of pestivirus on the dairy sector, perhaps because the impacts of pestivirus are more easily measurable in dairy than in beef production enterprises. An example of this fact is that it costs more to take an individual blood sample from a cross section of a beef herd to determine herd exposure to pestivirus than taking a single sample from the milk vat from a dairy farm.
The reproductive model used on the farm also has an important impact on the persistence of pestivirus within the herd. Herds that undertake year-round calving tend to have more prolonged episodes of exposure to the virus than seasonal or split calving herds, as there is a higher chance of a persistently infected (PI) animal being present in the herd.
It is estimated that approximately 5% of adult dairy cows will become infected with pestivirus each year in seasonal calving herds that do not undertake any virus control measures and around 2-3% of these infections will occur within the high-risk breeding phase, but not all of these cows will become pregnant.
Nevertheless, across dairy herds with no pestivirus control measures, herds that become infected will experience impacts of the virus on reproductive performance, milk production, cow immunity and calf health compared to dairy herds that are not exposed to the virus.
It’s difficult for farmers to quantify the effect of pestivirus in endemically infected herds due to its impact on many different aspects in the production cycle. Immune suppression, which presents as increased disease events and increased severity of disease is not often attributed to pestivirus. In dairy herds, pestivirus induced immune suppression and tissue damage has been associated with calf scours, pinkeye, mastitis, lameness and pneumonia. Combined these represent the most economically significant diseases in the dairy industry, therefore exacerbating these diseases will have a large downside on dairy farms.
In dairies, there have been severe outbreaks of pestivirus, with multiple PI calves being born over a short time horizon. On many occasions this has occurred when susceptible dairy herds have been exposed to the virus at their most vulnerable stage of the breeding cycle. As with beef herds, this often occurs with introductions to the herd in expansion or recovery phases. Pestivirus has also been introduced with heifers bred away on agistment or taken to dairy shows while pregnant.
Where a high proportion of the dairy cows are identified as susceptible to pestivirus, important strategies are to:
- Implement a vaccination program as a primary risk management tool
- Maintain a high-level biosecurity to limit the chance of exposure and importing the virus onto farm
- Testing should be conducted as part of a regular herd monitoring process
Pestivirus in the feedlot
The incidence of pestivirus in the feedlot sector is relatively common with studies showing that within the first six weeks of entry to a feedlot nearly 70% of susceptible cattle become infected with the virus. Of these cattle that do become infected, their risk of developing bovine respiratory disease (BRD) is 70% higher than uninfected cattle. Despite this, a cost:benefit of rectifying this situation and implementing mitigation strategies has not been determined.
By the time cattle are turned off from the feedlot it was estimated that just 6% of the cattle exiting the feedlot remained unexposed to the virus. Given the absence of the reproductive cycle at the feedlot the impact of pestivirus infection can often be overshadowed by other factors impacting the success of the cattle fattening process.
However, the adoption of pestivirus control measures in breeding herds still have a benefit for the feedlot sector as fewer PI animals present in the feedlot will likely reduce losses from respiratory diseases (such as Bovine Respiratory Disease – BRD) or from other viruses, that are enhanced in cattle with compromised immune systems from a pestivirus infection.
Multiple Australian studies have shown that there is a link between pestivirus and BRD however, there is a lack of consensus within the industry as to how to manage it. Most of this is driven by lack of appropriate cost:benefit calculations for controlling pestivirus in Australian feedlots.
The EP3 team have planned a follow up article that will investigate control/eradication programs being undertaken overseas to effectively manage the virus and what can be done domestically to improve upon current management processes.
- McGowan, M., et al., Epidemiology and Management of BVDV in Rangeland Beef Breeding Herds in Northern Australia. Viruses, 2020. 12(10).